Extend the gift of longevity.
At Longevity by Pure Global Ventures, we operate at the intersection of US-managed clinical oversight and Colombian biotechnological mastery. Our regenerative protocols utilize exclusively culture-expanded Mesenchymal Stem Cells (MSCs) processed in facilities certified by INVIMA (Colombia’s equivalent to the FDA). We do not settle for minimal manipulation; we demand cellular supremacy.


While US regulations heavily restrict stem cell expansion (limiting treatments to low-yield, same-day centrifuges), Colombia’s INVIMA provides a stringent, legal, and highly regulated framework for true cellular expansion. This allows our partner laboratories to multiply cells over several days in ISO-certified cleanrooms, delivering millions of highly potent, targeted MSCs safely and legally.
✔️ INVIMA Certified: Good Elaboration Practices (BPE).
✔️ ISO 9001 / ISO 13485: Global standards for medical devices and quality management.
✔️ US-Managed Protocols: Oversight by our US-based medical board.
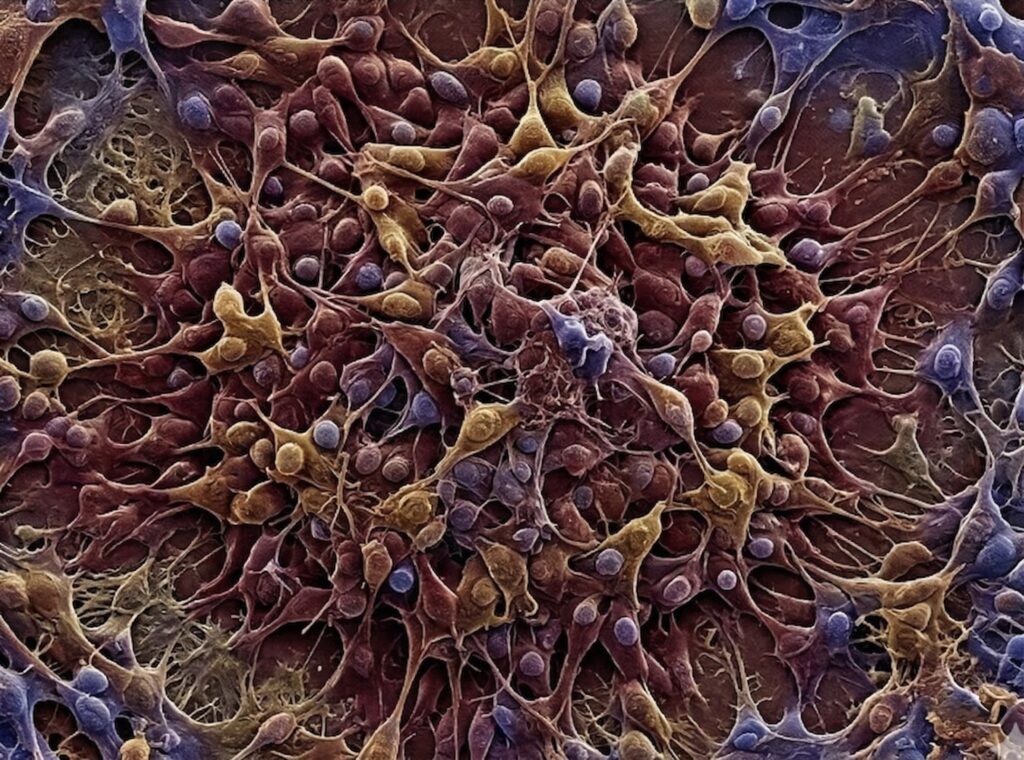
We guarantee 100M to 300M culture-expanded MSCs, achieving therapeutic impact impossible with standard U.S. procedures.
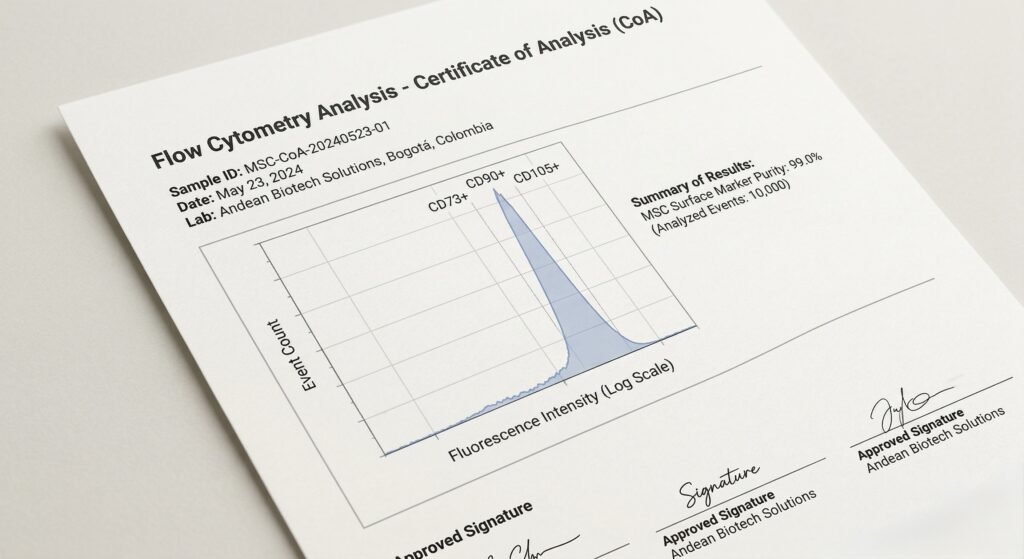
Verified via Flow Cytometry to ensure >95% expression of CD73, CD90, and CD105 markers with zero impurities.
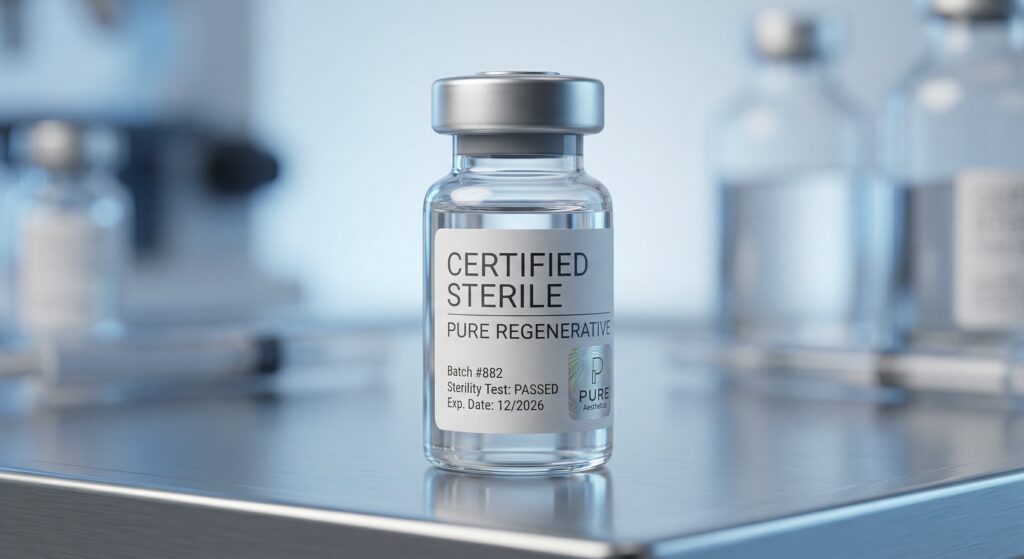
Strict third-party testing for bacteria and fungi. Tissues sourced exclusively from pre-screened, INVIMA-cleared donors.